x
k
l
a
r
c
h
e
m
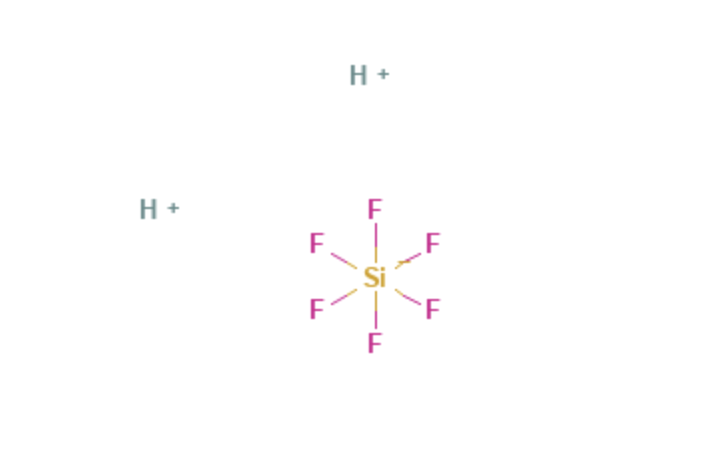
Hexafluorosilicic Acid
Chemical Product
Hexafluorosilicic Acid
Physical and Chemical Properties:
| Properties | |
|---|---|
| Appearance | Colorless to pale yellow liquid |
| Odor | Odorless |
| Molecular Formula | H2SiF6 |
| Molecular Weight | 144.1 g/mol |
| Boiling Point | 130-140 °C |
| Density | 1.33 g/cm³ |
| Solubility | Soluble in water |
| Flash Point | Not applicable |
| Chemical Stability | Stable under normal conditions but can react with metals and strong oxidizing agents |
Hexafluorosilicic acid is an inorganic compound used mainly in water fluoridation, in the production of aluminum, and in various chemical processes.
- Water Fluoridation: Used in municipal water systems for fluoridation.
- Aluminum Production: Used as a flux in the production of aluminum and aluminum alloys.
- Chemical Manufacturing: Used in the production of fluorosilicic acid salts and in other fluorine-containing chemicals.
- Industrial Applications: Used in the manufacture of semiconductors and in the etching of glass.
Physical and Chemical Properties:
| Properties | Data |
|---|---|
| Appearance | Colorless to pale yellow liquid |
| Odor | Odorless |
| Molecular Formula | H2SiF6 |
| Molecular Weight | 144.1 g/mol |
| Boiling Point | 130-140 °C |
| Density | 1.33 g/cm³ |
| Solubility | Soluble in water |
| Flash Point | Not applicable |
| Chemical Stability | Stable under normal conditions but can react with metals and strong oxidizing agents |