x
k
l
a
r
c
h
e
m
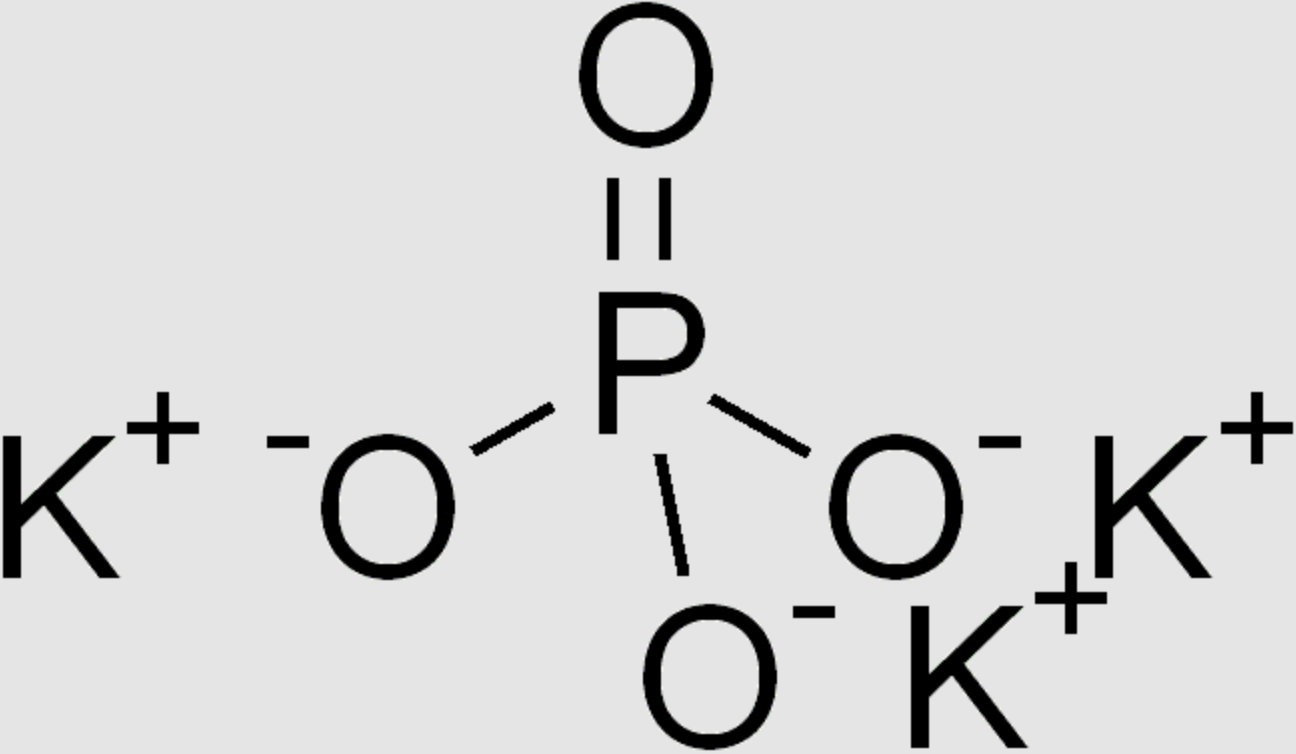
Potassium Tripolyphosphate
Chemical Product
Potassium Tripolyphosphate
Physical and Chemical Properties:
| Properties | |
|---|---|
| Appearance | White crystalline solid |
| Odor | Odorless |
| Molecular Formula | K3PO4 |
| Molecular Weight | 212.26 g/mol |
| Melting Point | 1336 °C |
| Density | 2.564 g/cm³ |
| Solubility | Soluble in water |
| Flash Point | Not applicable |
| Chemical Stability | Stable under normal conditions, but should be stored in a tightly closed container away from strong acids |
Potassium Tripolyphosphate is a water-soluble inorganic compound used primarily as a dispersing agent, water treatment chemical, and in industrial applications.
- Water Treatment: Used in water softening and in the removal of calcium and magnesium ions.
- Detergents: Applied as a component in detergents to improve cleaning efficiency.
- Chemical Manufacturing: Used as a dispersing agent in the formulation of fertilizers and other chemical products.
- Food Industry: Used as a sequestrant and emulsifier in food products.
Physical and Chemical Properties:
| Properties | Data |
|---|---|
| Appearance | White crystalline solid |
| Odor | Odorless |
| Molecular Formula | K3PO4 |
| Molecular Weight | 212.26 g/mol |
| Melting Point | 1336 °C |
| Density | 2.564 g/cm³ |
| Solubility | Soluble in water |
| Flash Point | Not applicable |
| Chemical Stability | Stable under normal conditions, but should be stored in a tightly closed container away from strong acids |