x
k
l
a
r
c
h
e
m
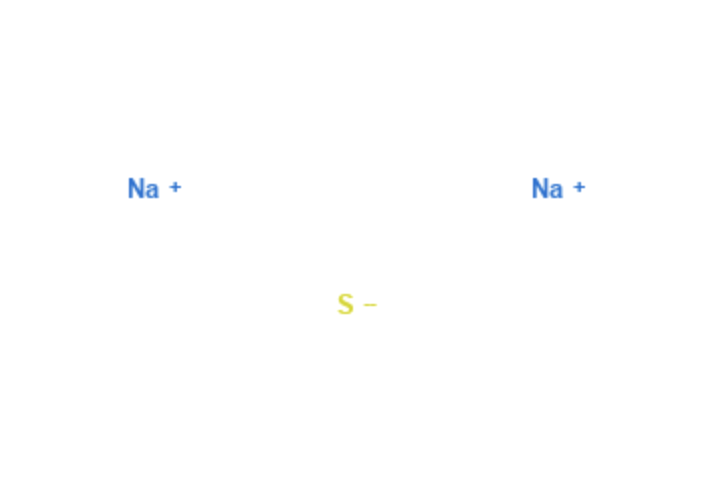
Sodium Sulfide
Chemical Product
Sodium Sulfide
Physical and Chemical Properties:
| Properties | |
|---|---|
| Appearance | Yellow to brown solid |
| Odor | Characteristic sulfurous odor |
| Molecular Formula | Na2S |
| Molecular Weight | 78.04 g/mol |
| Melting Point | 1180 °C |
| Density | 1.856 g/cm³ |
| Solubility | Soluble in water |
| Flash Point | Not applicable |
| Chemical Stability | Stable under normal conditions, but reacts with water and acids |
Sodium Sulfide is an inorganic compound used primarily in the manufacture of chemicals, water treatment, and in the leather industry.
- Chemical Manufacturing: Used in the production of other chemicals like sodium thiosulfate and sodium hydrosulfide.
- Water Treatment: Applied in water treatment as a reducing agent and to remove heavy metals.
- Leather Industry: Used in the leather tanning process to remove hair from hides.
- Pulp and Paper Industry: Used as a cooking chemical in the kraft process for making paper pulp.
Physical and Chemical Properties:
| Properties | Data |
|---|---|
| Appearance | Yellow to brown solid |
| Odor | Characteristic sulfurous odor |
| Molecular Formula | Na2S |
| Molecular Weight | 78.04 g/mol |
| Melting Point | 1180 °C |
| Density | 1.856 g/cm³ |
| Solubility | Soluble in water |
| Flash Point | Not applicable |
| Chemical Stability | Stable under normal conditions, but reacts with water and acids |