x
k
l
a
r
c
h
e
m
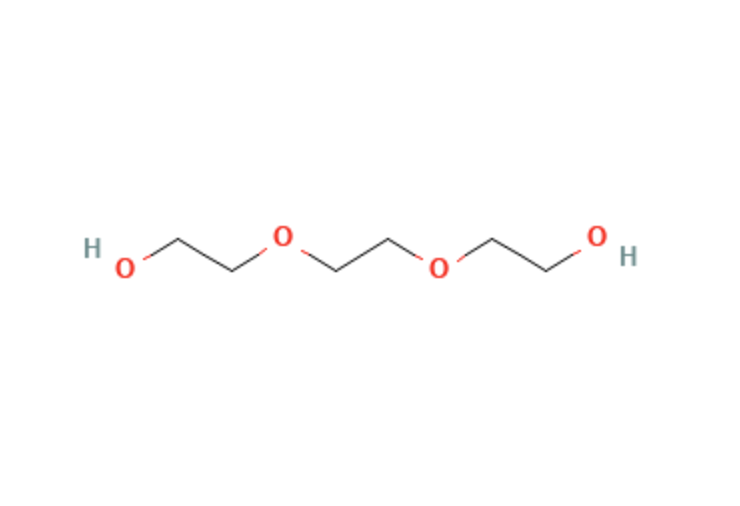
Triethylene Glycol
Chemical Product
Triethylene Glycol
Physical and Chemical Properties:
| Properties | |
|---|---|
| Appearance | Colorless, odorless liquid |
| Odor | Odorless |
| Molecular Formula | C6H14O3 |
| Molecular Weight | 150.19 g/mol |
| Boiling Point | 245 °C |
| Density | 1.126 g/cm³ |
| Solubility | Miscible with water and most organic solvents |
| Flash Point | 180 °C |
| Chemical Stability | Stable under normal conditions, but should be kept away from strong acids and bases |
Triethylene glycol is an organic compound commonly used as a solvent in industrial applications, as a humectant in cosmetics, and as an antifreeze agent.
- Chemical Manufacturing: Used as a solvent in paints, coatings, and adhesives.
- Pharmaceuticals: Applied in the production of pharmaceutical formulations and as a carrier solvent.
- Cosmetics: Used as a humectant and stabilizer in personal care products.
- Industrial Applications: Used as a heat transfer fluid and in antifreeze solutions.
Physical and Chemical Properties:
| Properties | Data |
|---|---|
| Appearance | Colorless, odorless liquid |
| Odor | Odorless |
| Molecular Formula | C6H14O3 |
| Molecular Weight | 150.19 g/mol |
| Boiling Point | 245 °C |
| Density | 1.126 g/cm³ |
| Solubility | Miscible with water and most organic solvents |
| Flash Point | 180 °C |
| Chemical Stability | Stable under normal conditions, but should be kept away from strong acids and bases |